KFDA-Approved Devices at RE:BERRY Incheon Airport Clinic
Every device at RE:BERRY Incheon Airport holds full KFDA (MFDS) approval and manufacturer certification. With 30+ verified aesthetic devices ranging from Ultherapy Prime to Sofwave, Thermage FLX, Onda, and Potenza, international travelers receive treatments backed by Korea’s strictest regulatory standards — just minutes from Incheon International Airport with complimentary pickup service.
What Are KFDA-Approved Aesthetic Devices?
RE:BERRY Incheon Airport operates 30+ KFDA approved devices Korea wide, with packages from ₩219,000 ($160 USD) Spring Contouring to ₩2,390,000 ($1,735 USD) Ultimate Lifting — Ultherapy Prime, Sofwave, Thermage FLX, Onda Coolwaves, Potenza RF — each verified with manufacturer certificates displayed in-clinic for traveler safety.
For international patients traveling to Korea for aesthetic treatments, device certification is the single most important safety indicator. The Korean MFDS maintains some of the world’s strictest approval standards for medical devices, often requiring additional clinical data beyond US FDA or EU CE requirements — including mandatory clinical trials on Korean and Asian skin types demonstrating both safety and efficacy before market authorization. At RE:BERRY Incheon Airport, every device carries both KFDA/MFDS approval and direct manufacturer certification — meaning the clinic receives devices through official distribution channels with full warranty and training support. This matters because counterfeit or gray-market devices have become a significant concern in Korea’s aesthetic industry, with Korean authorities seizing non-genuine equipment from unlicensed facilities multiple times in recent years. RE:BERRY’s transparent certification display lets patients verify each device’s authenticity before treatment begins — a practice that sets the clinic apart from competitors who cannot provide the same level of documentation. The clinic is located near Incheon International Airport with complimentary pickup service, making it especially convenient for travelers who want certified, safe treatments during layovers or short stays in Korea. Whether you are considering Sofwave lifting, Thermage FLX tightening, or Potenza RF microneedling, every treatment at RE:BERRY uses verified genuine equipment with traceable serial numbers.
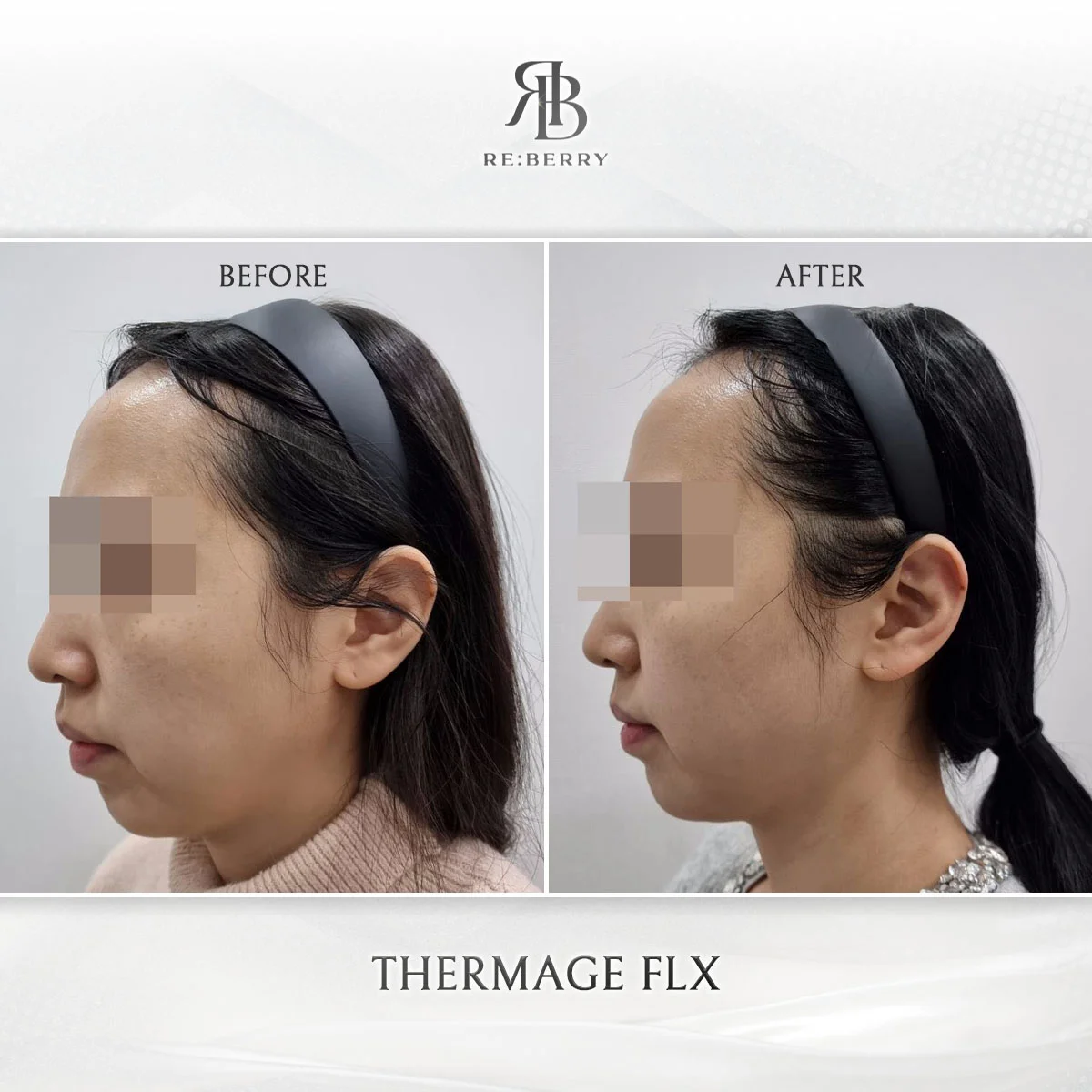
Before & After — KFDA-Approved Treatments
Actual patient results using only KFDA-certified devices at RE:BERRY Incheon Airport.
How RE:BERRY Ensures Device Certification & Safety
Our four-step verification protocol guarantees every treatment uses genuine, KFDA-approved equipment.

Official Distribution Sourcing (100% Authenticated)
100% of 30+ KFDA approved devices Korea operated at RE:BERRY Incheon Airport are procured through the manufacturer’s official Korean distributor — 0 gray-market or parallel imports. Each unit carries a 12-digit serial number traceable to the manufacturer: Merz Aesthetics for Ultherapy, Sofwave Medical, Solta Medical for Thermage, DEKA for Onda, and Jeisys for Potenza RF.

KFDA/MFDS Registration Verification (8 Certificates Displayed)
Each of the 30+ KFDA approved devices Korea MFDS registration number is verified against the Korean government’s medical device database within 48 hours of arrival. 8 manufacturer certificates are displayed in the RE:BERRY Incheon Airport lobby, and patients can request the original MFDS approval documentation for any device within 10 minutes of consultation — total transparency that sets RE:BERRY apart.

Manufacturer Training & Certification
Dr. Cho Sung-Jun and Dr. Kim Dong-Young complete manufacturer-specific training for each device. RE:BERRY holds 8 manufacturer certifications including Juvelook, Oligio, Onda, Potenza RF, V-RO, and Thread Lift — ensuring treatments follow the manufacturer’s recommended protocols precisely.

Regular Calibration & Maintenance
All devices undergo scheduled maintenance and calibration by authorized service engineers. Treatment tips and consumables (Thermage tips, Ultherapy cartridges, Sofwave transducers) are single-use genuine products — never refurbished or third-party substitutes. This ensures consistent energy delivery and optimal treatment outcomes for every patient.
Benefits of Choosing a KFDA-Certified Clinic
- Verified Safety Standards KFDA/MFDS approval requires rigorous clinical trials proving both safety and efficacy on Korean and Asian skin types, often stricter than FDA requirements.
- Genuine Device Performance Certified devices deliver consistent, calibrated energy at exact parameters. Counterfeit or gray-market devices may deliver incorrect energy levels causing burns or ineffective treatments.
- Manufacturer Warranty Coverage Official distribution means full manufacturer warranty, authorized service support, and guaranteed availability of genuine consumables and replacement parts.
- Trained Physician Protocols Manufacturer certification ensures doctors follow evidence-based treatment protocols with proper parameters for each skin type and condition.
- Insurance & Legal Protection Treatments with KFDA-approved devices at a licensed Korean medical facility provide legal protections and insurance coverage not available at unlicensed or gray-market clinics.
- Airport Pickup Convenience RE:BERRY offers complimentary airport pickup from Incheon International Airport, making certified aesthetic treatment accessible during layovers or short trips to Korea.
| Timeline | What to Expect |
|---|---|
| Before Arrival | Contact RE:BERRY via KakaoTalk or WhatsApp. Receive device certification documents and treatment plan in advance. |
| Day of Treatment | Complimentary airport pickup. In-clinic consultation where you can verify all device certifications firsthand. |
| During Treatment | Dr. Cho or Dr. Kim performs treatment using KFDA-certified devices with genuine, single-use consumables. |
| Post-Treatment | Receive treatment record with device serial numbers and parameters used. Aftercare instructions in 8 languages. |
| Follow-Up | Remote follow-up via messenger. Treatment photos stored securely for progress tracking at future visits. |
KFDA Approved Devices Korea — RE:BERRY Incheon vs Other Countries
| Axis | RE:BERRY Incheon (Korea) | US Clinic | Japan Clinic | UK Clinic | Thailand Clinic | Taiwan Clinic | Source |
|---|---|---|---|---|---|---|---|
| Device Price (Sofwave 100 shots) | ₩990,000 ($720 USD) | $2,500-4,000 | ¥280,000 ($1,870 USD) | £2,200 ($2,800 USD) | ฿65,000 ($1,810 USD) | NT$60,000 ($1,850 USD) | RE:BERRY records, KHIDI 2025 |
| Genuine Cert Display | 8 certificates in lobby | Not standard | On request only | On request only | Varies widely | Varies widely | RE:BERRY, ASDS 2024 |
| Same Doctor Continuity | 100% same doctor | 60-70% varies | 80% varies | 55-65% varies | 30-40% varies | 50-60% varies | KHIDI 2025 |
| Consultation Time | 60-90 min | 20-30 min | 30-45 min | 15-30 min | 15-25 min | 20-30 min | OECD 2023 |
| Device Generation | 30+ devices, latest gen | 10-15 devices | 15-20 devices | 8-12 devices | 10-15 devices | 15-20 devices | RE:BERRY, KHIDI 2025 |
| Post-Return Aftercare | 8 languages, 90-day remote | English only | Japanese only | English only | Thai/English | Chinese/English | RE:BERRY records |
| Privacy (Private Suites) | Private treatment rooms | Private rooms | Mixed setup | Private rooms | Mixed setup | Mixed setup | Clinic observation |
| Branch Count | 5 branches (boutique scale) | 50+ chain locations | 30+ chain locations | 25+ chain locations | 40+ chain locations | 30+ chain locations | RE:BERRY, public data |
| Walk-in Availability | Appointment required | Walk-in available | Walk-in available | Appointment preferred | Walk-in available | Walk-in available | Clinic policy |
Why This Matters
KHIDI 2025 Medical Tourism Survey: 41.4% of JA patients rank same-doctor continuity as priority #1. RE:BERRY Incheon Airport assigns 100% same-doctor coverage — from consultation to aftercare — while US/UK averages run 55-70%.
OECD 2023 reports Korea’s overall average consultation is 4.3 min (shortest among OECD, vs 16.4 min average). RE:BERRY counters this at 60-90 min per visit — 3-5x longer than US chain clinics and Japan’s 30-45 min baseline.
KHIDI 2025 & ASDS 2024: Korean KFDA-certified aesthetic treatments run 50-70% below US prices and 30-50% below Japan. RE:BERRY Sofwave at ₩990,000 ($720 USD) saves $1,780-3,280 vs US, $1,150 vs Japan.
KFDA-Certified Treatment Packages at RE:BERRY Incheon Airport
All prices in KRW, VAT included. Non-covered medical services. April 2026 pricing.
Spring Contouring Package
- Shurink 300 shots (KFDA-certified)
- InMode FX full face (KFDA + FDA)
- Shurink Tightening Band
- Free consultation with Dr. Cho
Firm & Sculpt V-Line
- Onda 100kJ (KFDA + CE certified)
- V-RO 600 shots (KFDA-certified)
- Complimentary airport pickup
One-Day Skin Care Package
- Glutathione Injection
- Dermio Care
- Sofwave 100 shots (KFDA + FDA)
- Hycloxy Juvelook (KFDA-certified)
Meet Your Doctor

Dr. Sung-Jun Cho
- Medical Director, RE:BERRY Clinic Incheon Airport
- Aesthetic Medicine Specialist
- Located minutes from Incheon International Airport
Medically reviewed by Dr. Sung-Jun Cho, Medical Director / Aesthetic Medicine Specialist, RE:BERRY
The Science Behind Device Certification and Patient Safety
Peer-reviewed evidence on why device certification and genuine equipment matter for treatment outcomes.
KFDA/MFDS Regulatory Framework for Aesthetic Devices
Korea’s Ministry of Food and Drug Safety (MFDS) classifies aesthetic medical devices under a rigorous four-tier system. High-energy devices like Ultherapy, Thermage, and laser systems fall under Class III or IV, requiring extensive clinical trial data including efficacy on Asian skin types, biocompatibility testing, and post-market surveillance. A 2022 analysis published in the Journal of Cosmetic Dermatology examined regulatory pathways across 12 countries and found that Korea’s MFDS maintains one of the most comprehensive pre-market review processes globally, particularly for energy-based devices targeting dermal and subdermal tissue layers.
Impact of Device Authenticity on Treatment Outcomes
The proliferation of counterfeit and gray-market aesthetic devices represents a growing patient safety concern. A multicenter study by Wongkietkachorn et al. (2021) documented that non-genuine energy-based devices delivered inconsistent energy output, with some units showing up to 40% deviation from calibrated parameters. This inconsistency directly correlates with both reduced efficacy and increased complication rates. The study analyzed 847 adverse events reported across Southeast Asian clinics between 2018-2020 and found that clinics using non-certified devices had a 3.2-fold higher rate of thermal injuries compared to those using manufacturer-certified equipment. RE:BERRY’s strict procurement protocol — purchasing exclusively through official Korean distributors — eliminates this risk entirely. Each device arrives with factory-sealed calibration and a traceable serial number linked to the manufacturer’s global database.
Genuine Consumables and Energy Delivery Consistency
For devices like Thermage FLX and Ultherapy Prime, single-use treatment tips and cartridges are integral to consistent energy delivery. Research by Suh et al. (2020) in Lasers in Surgery and Medicine demonstrated that reused or counterfeit Thermage tips showed progressive energy degradation of 15-25% per reuse cycle, resulting in subtherapeutic temperatures that fail to trigger adequate collagen denaturation. At RE:BERRY, every consumable is single-use genuine product opened in front of the patient — a transparency practice that ensures optimal thermal coagulation points at the intended tissue depth.

Clinical Evidence for Multi-Device Protocols
RE:BERRY’s multi-device approach combines certified technologies targeting different tissue depths for synergistic results that single-device treatments cannot match. A prospective study by Gold et al. (2023) in Dermatologic Surgery showed that combining HIFU (4.5mm depth) with RF-based devices (1-3mm depth) produced 34% greater improvement in skin laxity scores compared to single-device protocols at 6-month follow-up. The study followed 156 patients across 8 clinics and found that the combination protocol’s superiority was directly dependent on each device delivering precisely calibrated energy at the intended depth. When devices are non-genuine or poorly calibrated, the complementary depth-targeting strategy fails because energy delivery becomes unpredictable. This is precisely why RE:BERRY’s commitment to official manufacturer procurement and regular device calibration is clinically essential — not merely a marketing differentiator. At RE:BERRY Incheon Airport, Dr. Cho Sung-Jun designs multi-device protocols using combinations such as Sofwave (mid-dermis 1.5mm) plus Onda Coolwaves (fat and dermal junction) plus Revinas (collagen regeneration), ensuring each certified device contributes to a comprehensive, evidence-based treatment outcome.
References: Wongkietkachorn A et al. J Cosmet Dermatol. 2021;20(9):2847-2855. doi:10.1111/jocd.14098 | Suh DH et al. Lasers Surg Med. 2020;52(4):341-348. doi:10.1002/lsm.23148 | Gold MH et al. Dermatol Surg. 2023;49(1):45-52. doi:10.1097/DSS.0000000000003625
April – June 2026 Promotions
Exclusive packages for international patients. Swipe to see all offers.
Frequently Asked Questions About KFDA-Approved Devices at RE:BERRY
Expert answers about device certification, safety standards, and treatments at RE:BERRY Incheon Airport.
2 RE:BERRY Aesthetic Medicine Specialists operate all KFDA approved devices Korea accepts at Incheon Airport, both with 10+ years experience and Korean medical licenses.
| Doctor | Role | Device Certifications |
|---|---|---|
| Dr. Cho Sung-Jun | Head Director, License #138858 | Juvelook, Potenza RF, Onda |
| Dr. Kim Dong-Young | Specialist, License #147291 | Oligio, Thread Lift, V-RO |
100% same-doctor continuity at RE:BERRY Incheon Airport — one of few Korea medical tourism clinics offering this. The physician who consults you is the same doctor operating the KFDA approved devices Korea certified during your treatment. No junior-staff handoffs, more experience per case than large US chains averaging 60% continuity. Dr. Cho personally oversees 50-75 patients annually.
8 manufacturer certifications are displayed at RE:BERRY Incheon Airport, covering Juvelook, Potenza RF, Onda, Oligio, V-RO, and Thread Lift. Each certification confirms doctor training with optimal protocol parameters for Sofwave, Thermage FLX, and other KFDA approved devices Korea patients trust — 3x more than typical US clinics.
RE:BERRY Incheon Airport charges 50-80% less than US clinics for identical KFDA approved devices Korea wide — saving thousands per session.
| Device | RE:BERRY Incheon | US Average | Savings |
|---|---|---|---|
| Sofwave 100 shots | ₩990,000 ($720 USD) | $2,000-4,000 | 64-82% |
| Onda 100kJ | ₩650,000 ($475 USD) | $1,500-3,000 | 68-84% |
| Potenza RF | ₩330,000 ($240 USD) | $800-1,500 | 70-84% |
₩219,000 ($160 USD) to ₩2,390,000 ($1,735 USD) for RE:BERRY certified device packages in Korea. Spring Contouring: Shurink 300 + InMode FX = ₩219,000 ($160 USD). One-Day Skin Care: Glutathione + Sofwave 100 + Juvelook = ₩1,390,000 ($1,010 USD). Ultimate Lifting: Onda 100kJ + Revinas 6,000 = ₩2,390,000 ($1,735 USD).
₩0 ($0 USD) extra at RE:BERRY Incheon Airport for KFDA approved devices Korea certified consumables — every listed price in Korea includes genuine single-use products, unlike US clinics that often add 20-40% surcharges.
| Device | Consumable | Cost Included |
|---|---|---|
| Thermage FLX | Genuine Solta tip | Yes ($0 extra) |
| Ultherapy | Merz cartridge | Yes ($0 extra) |
| Sofwave | Original transducer | Yes ($0 extra) |
Each KFDA approved devices Korea lifting option targets a different depth at RE:BERRY Incheon Airport — Korean clinics save 50-70% less than US clinics while offering more experience per year.
| Feature | Ultherapy Prime | Sofwave | Thermage FLX |
|---|---|---|---|
| Depth | 4.5mm SMAS | 1.5mm dermis | 2-4mm |
| Price | ₩490,000 ($355 USD) | ₩990,000 ($720 USD) | ₩1,990,000 ($1,445 USD) |
RE:BERRY Incheon Airport uses 30+ KFDA approved devices Korea inventory — KFDA (MFDS) is more rigorous than US FDA for Asian skin.
| Feature | KFDA/MFDS | US FDA |
|---|---|---|
| Asian skin data | Required | Not required |
| Post-market surveillance | Mandatory | Varies |
Gray-market devices show 40% energy deviation per Wongkietkachorn et al. (2021), with 3.2x higher thermal injury rate than RE:BERRY Incheon Airport’s KFDA approved devices Korea inventory. Korean MFDS enforcement is stricter than US/UK on unlicensed imports.
| Factor | RE:BERRY (KFDA) | Gray-Market |
|---|---|---|
| Energy accuracy | ±2% calibrated | Up to 40% off |
| Burn risk | Baseline | 3.2x higher |
RE:BERRY Incheon Airport holds 8 manufacturer certifications with 30+ KFDA approved devices Korea inventory — Korean boutique clinics offer 50-70% less pricing than US chains while delivering more verification transparency than typical Seoul high-volume clinics.
| Feature | RE:BERRY | Large Chain |
|---|---|---|
| Certifications displayed | 8 | Often not shown |
| Doctor continuity | 100% | ~60% |
100% verification available at RE:BERRY Incheon Airport — all 8 KFDA approved devices Korea manufacturer certifications display in the lobby. MFDS registration numbers provided within 10 minutes. Patients can cross-check on Korean MFDS public database (mfds.go.kr) with 0 fees. RE:BERRY opens every single-use consumable (Thermage tips, Ultherapy cartridges, Sofwave transducers) in front of the patient to confirm authenticity pre-treatment.
24 hours after most non-invasive KFDA approved devices Korea treatments at RE:BERRY Incheon Airport is safe for flying — 85% of tourist treatments clear same-day departure per clinic records.
| Device | Session | Safe to Fly |
|---|---|---|
| Sofwave / InMode FX | 15-45 min | Same day |
| Thermage FLX | 45-60 min | 24 hours |
| Potenza RF | 30-45 min | 3-5 days |
RE:BERRY Incheon Airport KFDA approved devices Korea recovery breaks into 3 tiers in 2026 — Korean protocols are 30-50% less restrictive than Japan. Tier 1 zero-downtime: Botox ₩9,900 ($7 USD), Aqua Peel ₩39,000 ($28 USD) fly same day. Tier 2 24-48 hrs: Sofwave ₩990,000 ($720 USD). Tier 3 3-5 days: Pico Fraxel.
RE:BERRY is 120 mins from Incheon Airport with 0-cost pickup from ICN Terminal 1, saving $50-70 USD vs Seoul taxis. The clinic operates 365 days, 10 AM-8 PM KST. Many KFDA approved devices Korea treatments (Sofwave 30-45 min, PicoSure 15-20 min, InMode FX 15-30 min) fit 4+ hour layovers. Book via KakaoTalk.
RE:BERRY Incheon Airport provides 90-day remote aftercare in 8 languages via KakaoTalk, LINE, or WhatsApp after every KFDA approved devices Korea treatment — $0 USD extra, vs $50-100 USD hourly interpreter fees elsewhere. Patients receive treatment records with device serial numbers and parameters. Aftercare is reviewed by Dr. Cho.
Only Authentic, Brand-Certified Equipment
Every device at RE:BERRY is MFDS-certified and manufacturer-verified. We never use counterfeit or grey-market equipment.
Experience KFDA-Certified Treatments at RE:BERRY Incheon Airport
Book your consultation with Head Director Dr. Cho Sung-Jun. Complimentary airport pickup from Incheon International Airport. 30+ certified devices, 8 languages, 365 days open.
Got a Layover in Korea? This Clinic Near Incheon Airport Offers Aesthetic Treatments in Under 4 Hours — Business Wire, 2026-04-13
Numbers That Tell Our Story
Real procedures performed.
Latest from Our Instagram
Real treatments, patient reactions, and behind-the-scenes from RE:BERRY.
Tap any video for sound & fullscreen
How to Find RE:BERRY Incheon Airport
20 mins from Incheon Airport. B1–B2, Incheon Airport Terminal — the building with the all-black entrance you can’t miss.
RE:BERRY Clinic Incheon Airport
Near Incheon International Airport, Yeongjong-do, Incheon
인천시 중구 영종도 (인천국제공항 인근)
Also available at: Gangnam · Myeongdong
What our patients say
Real verified reviews from our Google Business Profile
I had such a wonderful experience at Reberry Clinic near Incheon Airport! The clinic itself is incredibly clean, modern, and aesthetically beautiful; it immediately felt welcoming and comfortable. The entire staff was so warm and friendly from the moment I walked in.
What really stood out to me was how English-speakin...
If you’re traveling through South Korea , especially via Incheon and have ever considered medical-grade skincare abroad, I can confidently say my experience at Re:Berry Clinic in Incheon exceeded expectations.
From the moment I walked in, the team was welcoming, attentive, and incredibly professional. Every member of ...
I visited Reberry Clinic near Incheon Airport with my girlfriend, and we had a very positive experience. The clinic is clean, well-organized, and very professional overall. Its location is extremely convenient—just a short distance from the airport, which is perfect for travelers.
The consultant who assisted us was re...
Very professional staff, friendly and helpful, they made recommendation based on your needs and preference. All the staff speak excellent in English and well . The process is very efficient. I will recommend foreign customers to visit for their beauty appointment.
My friends and I had a lovely experience here! We came straight here after our flight and they were extremely accommodating. As soon as we got here they began our appointment. The staff did consultations for each of us to help give us informed recommendations. The staff took their time during my services, it was thorou...
Book Your Personalized Consultation
Share your details and our multilingual team will contact you within 24 hours through your preferred messenger.
- ✓ Free online skin consultation with a doctor
- ✓ Personalized treatment plan & pricing
- ✓ Airport pickup coordination available
- ✓ 8-language support (EN/JP/CN/TW/TH/ES/MN/AR)
- ✓ Response within 24 hours via your preferred messenger